In an additional study of adults with bipolar I or II depression,Observed change in depression symptoms in patients with mixed features, with or without symptoms of anxiety*, vs placebo1,2
Primary endpoint
MADRS total score from baseline
at 6 weeks in bipolar depression patients exhibiting mixed features
POST HOC analysis
MADRS total score in patients
with mixed features and symptoms of anxiety2‡
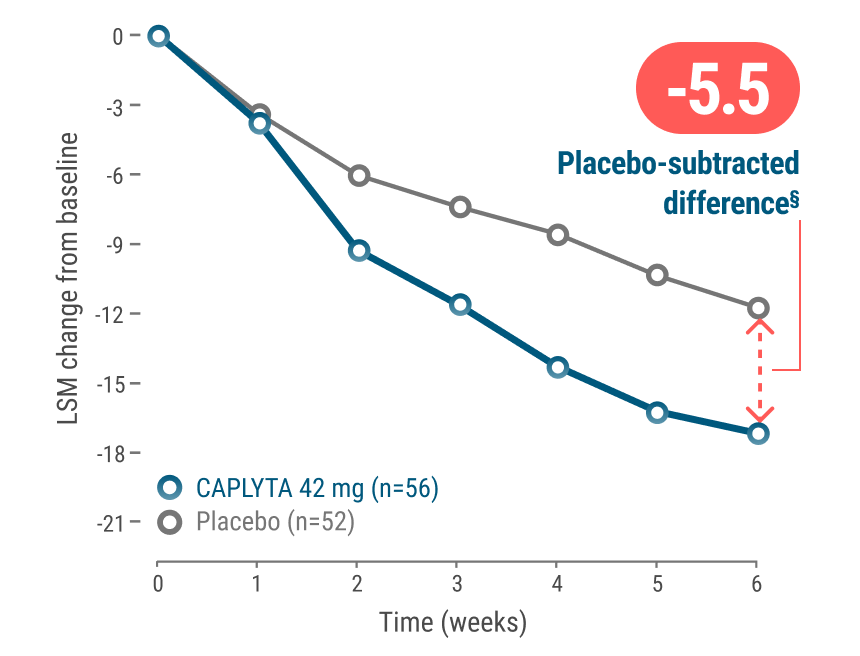
*CAPLYTA is not approved to treat mania or anxiety. Symptoms of anxiety in patients with bipolar depression were based on the DSM-5 criteria for anxious distress.2,4
†In patients with bipolar depression with mixed features, the placebo-subtracted difference was -5.7 for MADRS total score (95% CI: -8.3, -3.1). Mean baseline MADRS total scores for CAPLYTA 42 mg and placebo were 31.8 and 31.1 respectively.1
§Treatment difference is calculated as LSM of CAPLYTA minus LSM of placebo; all values are rounded.2
CI=confidence interval; LSM=least squares mean; MADRS=Montgomery-Åsberg Depression Rating Scale.
Mixed features & anxious distress criteria
As defined by the DSM-5, mixed features is defined by the presence of at least 3 of the following subsyndromal manic/hypomanic symptoms during most days of a depressive episode1:
- Elevated mood
- Inflated self-esteem
- Increased talkativeness
- Racing thoughts
- Increased risky behavior
- Increased energy or goal-directed activity
- Decreased need for sleep
As defined by the DSM-5, anxious distress is defined by the presence of ≥2 of the following symptoms during a depressive episode5:
- Feeling keyed up
- Feeling unusually restless
- Difficulty concentrating because of worry
- Fear that something bad may happen
- Feeling loss of control
CGI-S=Clinical Global Impression-Severity; DSM=Diagnostic and Statistical Manual of Mental Disorders; MADRS=Montgomery-Åsberg Depression Rating Scale; YMRS=Young Mania Rating Scale.





